BASIC REEF CHEMISTRY (PART I)
This is the first in two articles dealing with the basics of reef chemistry. This will be written as a crash course on what hobbyists need/ ought to know. The article to follow touches on information, which is useful but not necessary to know.
SALINITY
The salinity of seawater is typically given either in units of parts per thousand (PPT) or specific gravity (SG). The ideal range for salinity in a reef aquarium is usually placed between 33 to 38 PPT, which approximates to 1.024 to 1.028 SG. The middle ground of 35 PPT (or approximately 1.026 SG) is typically recommended and is the accepted “standard”.

Refractometer that measure PPT and SG level
PPT
PPT is a measure of the total weight of salt dissolved in seawater in relation to the total weight of the seawater reference. In the case of 35 PPT, this translates into 35g of salt in 1000g of seawater.
However, to evaporate 1000g of seawater each time to determine the salinity of a solution is obviously impractical. Therefore, there are other methods developed to determine the salinity of seawater.
Hydrometer that measure SG level
SG
SG measures the density of the object being measured relative to the density of a reference. For the purposes of the reef hobby, this refers to the density of seawater relative to the density of pure freshwater.
Since density is a measure of an object’s mass to its volume, and the volume of an object changes depending on its temperature, it is important to note that the density of seawater changes with temperature too. As a corollary, the SG of a sample of seawater also changes with temperature. For this reason, some devices used to measure salinity state particular temperatures at which samples should be measured at. This is especially important when using a hydrometer to measure the relative density of a sample of seawater.
How then does a refractometer measure salinity?
As with a hydrometer, a refractometer does not measure salinity directly. Instead, it is an instrument which measures the refractive index of a sample and correlates it to an SG value.
It is important to note the following when using a refractometer:
- Have the sample at 25 degree Celsius, or simply use a refractometer with automatic temperature control (ATC) which compensates for changes in temperature;
- Read the refractometer through a light source closest to daylight; and
- Use only a refractometer designed for seawater, not one that is designed for sugar, alcohol or brine.
PH
pH is the measure of concentration of hydrogen ions (H+) ions in a solution. The more acidic a solution is, the higher the concentration of H+ ions and the lower its value on the pH scale.
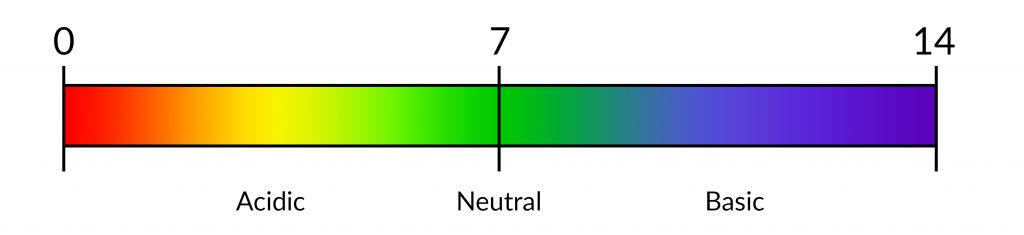
As can be seen above, pure water registers a pH of 7 on the scale.
It is important to pay attention to pH in a reef tank. A low pH is stressful to fish and can inhibit coral calcification. The recommended pH sits between 7.8 to 8.3.

nyos test kit
CALCIUM
The importance of Calcium, Magnesium and alkalinity in seawater cannot be understated.
Aside from Sodium and Chlorine (plus Hydrogen and Oxygen which form water itself), the most abundant elements in seawater are Calcium and Magnesium. In seawater, Calcium and Magnesium are dissolved in solution as Ca2+ and Mg2+ ions. The significance of this is a topic for another day. For now, it is sufficient to note that the amount of Magnesium dissolved in seawater has a bearing on the amount of Calcium that can be dissolved.
Calcium is needed for coral calcification. Hard corals use Calcium dissolved in seawater, together with carbonates present, as the primary building blocks for their skeletons. It is no surprise that this process is aptly termed “coral calcification”. Without Calcium, hard corals would not exist as they do today.
MAGNESIUM
Magnesium is the second most abundant cation in seawater. Its presence in seawater enables a higher concentration of Calcium ions than otherwise possible to be dissolved in seawater. Like Calcium, Magnesium is also incorporated by hard corals into their rigid skeleton.
ALKALINITY
Alkalinity completes the trinity of crucial components in seawater. Alkalinity is not an element. Instead, it describes the buffering capacity of water i.e. its ability to resist changes in pH.
In seawater, alkalinity is affected by two main systems. First, the carbonate buffer system. Second, and to a much lesser extent, the borate buffer system. This article focuses solely on carbonate buffers, save to state that while the presence of borate ions contributes to the alkalinity levels measured in seawater, it itself is not used for calcification.
Carbonate buffer (CO32-)
Seawater naturally contains carbonate ions (CO32-) and these exist in a chemical equilibrium with other ions. What this means is that carbonate ions, as one reactant, and some other ions, as other reactants, are both present at the same time. Whenever a reversible arrow (⇌) is used, think of it as a reaction that can go in both directions of the arrow.
When Carbon dioxide is dissolved in water, it partially dissociates in the following manner to exist in equilibrium with Carbonic acid:
Equation 1
CO2 + H2O ⇌ H2CO3
Carbonic acid itself can further dissociate to give H+ ions and bicarbonate ions:
Equation 2
H2CO3 ⇌ H+ + HCO3–
The bicarbonate ion also exists in an equilibrium with carbonate ions:
Equation 3
HCO3– ⇌ H+ + CO32-
If nothing is done to a solution containing ions in equilibrium, the ions do exactly what they suggest – maintain an equilibrium. However, if either side of any of the arrows above is disturbed, the equilibrium will shift to account for this change. In other words, a “buffer” against the change.
For example, if an acid is added:
- There will be more H+ ions in solution;
- The bicarbonate-carbonate equilibrium at Equation 3 shifts to the left to account for the H+ introduced and more HCO3– is present; and
- This process continues for Equations 1 and 2, with more carbonic acid and more Carbon dioxide resulting.
What eventually happens is that the concentration of H+ ions when added to a system containing a buffer does not increase the concentration of H+ ions in the total system as much as would have been the case if there were no buffers present. In effect, the system “neutralises” acids added. A titration curve of such a reaction would look roughly in the form of the sketch below.
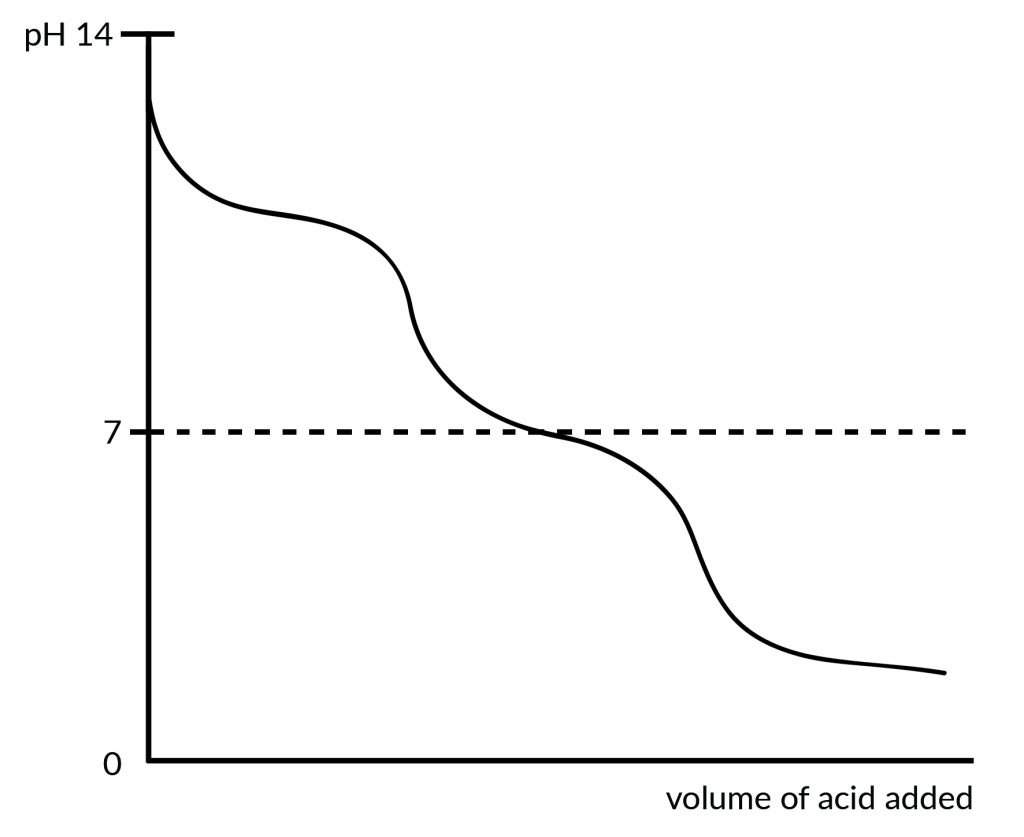
The same reasoning also applies if a base is added, although the Equations above will be pushed towards the right instead.
Importance of alkalinity
Maintaining the appropriate levels of alkalinity is important for three main reasons:
- It is a buffer against swings in pH;
- It is a good gauge for the amount of carbonates in seawater for corals to use for calcification; and
- It has an effect on the amount of Calcium ions that may be present. This will be explained in the second article to follow.
Do look out for Part 2 of the Reef chemistry ..